Epigenetics of Cell Differentiation
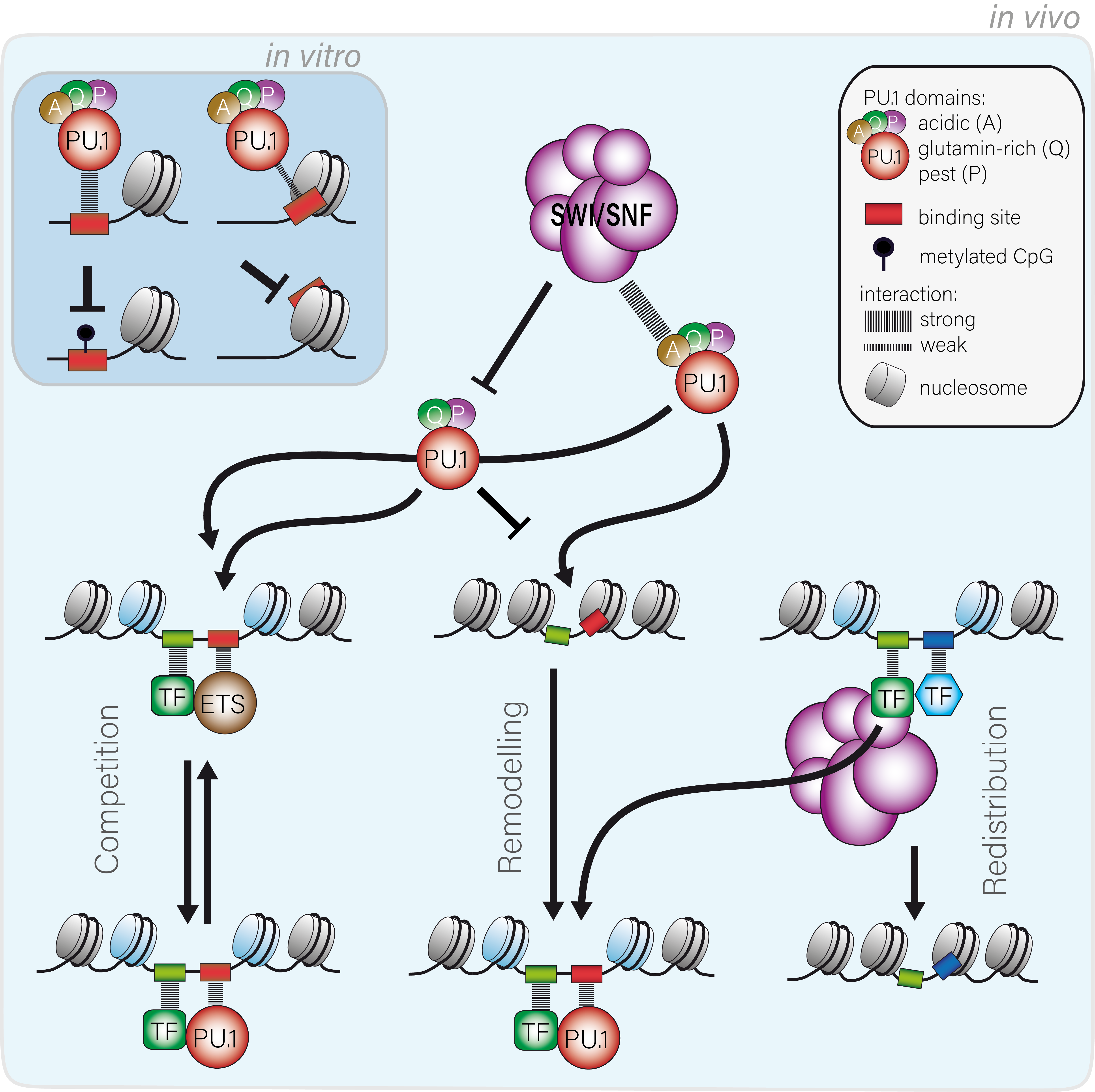
Cell type-specific gene regulation is controlled by the combination of specific transcription factors as well as epigenetic mechanisms that control the structure and accessibility of DNA in chromatin. Master regulators (also called ‘pioneering factors’) like the myeloid and B cell-specific transcription factor PU.1 play key roles in lineage differentiation and specification and share a unique ability to overcome chromatin restriction and to open up chromatin for other factors. However, the specific features of master regulators (or transcription factors in general) that enable them to access DNA in chromatin as well as the general rules for DNA binding (only a minor fraction of recognition sites is usually occupied) remain to be defined.
We recently showed that binding of the master regulator PU.1 to its binding sites in chromatin is controlled by a combination of sequence motif affinity, cooperativeness between neighboring binding sites and the engagement of chromatin remodeling complexes. The ability of PU.1 to shape regulatory landscapes and to act as a non-classic pioneer factor requires its N-terminal acidic activation domain and its interaction with the SWI/SNF remodeling complex. Interestingly, the redistribution of partner TFs by PU.1 also requires the SWI/SNF-interacting acidic domain, suggesting that the remodeler complex may act as part of a hub to collect and distribute co-associated TFs in a PU.1 dependent manner.
Michael Rehli • Dept. Internal Medicine III • University Hospital
F.-J.-Strauss Allee 11 • 93053 Regensburg • Germany
Imprint & Privacy